 
To the next page in the reactivity series sequence. The rest of the Reactivity Series topic looks at how you can build up this list, and its importance in chemistry. Common metals like aluminium and zinc and iron are somewhere in the middle. So sodium and potassium, for example, are extremely reactive, and gold is virtually unreactive. The reactivity series lists the metals (and a couple of non-metals) in order of decreasing reactivity. Along the side of the matrix, you should list the five metals used in this part of the experiment: copper, iron, magnesium, tin, and zinc. There will be much more detail in further pages in this section. An activity series of metals is a table of metals arranged in the order of their decreasing chemical activity or the ease at which the metal will give up. 
Do not proceed to schedule a custom demo unless you have already conferred with the lecture demonstrator about it.This page takes a brief look at what the reactivity series consists of. This lesson plan includes the objectives, prerequisites, and exclusions of the lesson teaching students how to use the reactions of metals with water. Purpose: The purpose of this experiment is to construct an activity series from some metallic elements. After observing or carrying out a series of reactions, you will construct an activity series for some elements. The custom demos section of the website is used by UO chemistry instructors to schedule demonstrations that are not listed in the database. An activity series allows one to make such predictions. The university shall not be liable for any special, direct, indirect, incidental, or consequential damages of any kind whatsoever (including, without limitation, attorney's fees) in any way due to, resulting from, or arising in connection with the use of or inability to use the web site or the content. The university further disclaims all responsibility for any loss, injury, claim, liability, or damage of any kind resulting from, arising out or or any way related to (a) any errors in or omissions from this web site and the content, including but not limited to technical inaccuracies and typographical errors, or (b) your use of this web site and the information contained in this web site.the university shall not be liable for any loss, injury, claim, liability, or damage of any kind resulting from your use of the web site. The ordering of the activity series can be related to the. 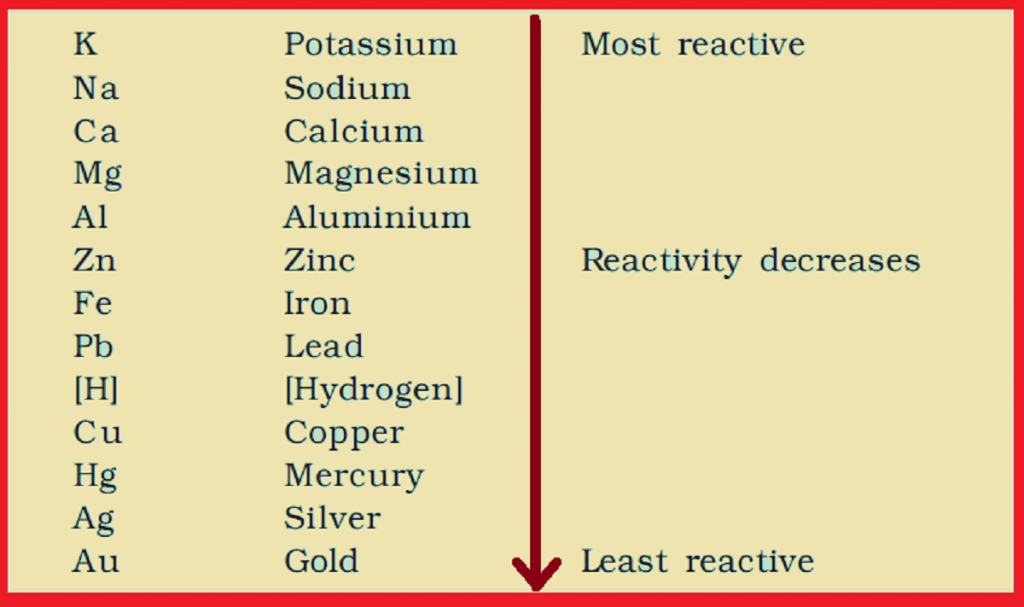
The starting materials are always pure elements, such as a pure zinc metal or hydrogen gas, plus an aqueous compound. In the reaction with a hydrogen-ion source, the metal is oxidized to a metal ion, and the hydrogen ion is reduced to H 2. A single replacement reaction, sometimes called a single displacement reaction, is a reaction in which one element is substituted for another element in a compound. Metals arranged in series are called activity series of metals because metals differ in tendency to lose valence electrons and can be arranged in a series. The university expressly disclaims all warranties, including the warranties of merchantability, fitness for a particular purpose and non-infringement. Depending on the ease with which the metals lose their electrons and form ions they are arranged in metal activity series or electrochemical series. The activity or electromotive series of metals is a listing of the metals in decreasing order of their reactivity with hydrogen-ion sources such as water and acids. Concept of the activity series: - Metals are elements with few valence electrons and these electrons are comparatively readily given up so that they form. This web site is provided on an "as is" basis. "Do not do demos unless you are an experienced chemist!" Please read the following disclaimer carefullyīy continuing to view the descriptions of the demonstrations you have agreed to the following disclaimer. The test tubes in each row are marked with a piece of colored tape corresponding to the color of tape on the beaker containing the strips of corresponding metal, e.g., the row of test tubes containing zinc nitrate solution and the beaker containing zinc strips might both be marked with blue tape.One beaker contains four copper strips.One beaker contains four silver strips.Four 125 mL beakers each containing four about 60 X 8 mm strips of a certain metal as follows:.The tubes on the fourth row contain 0.1 M zinc nitrate. The activity series of metals arranges all metals in order of their decreasing chemical activity.The tubes on the third row contain 0.1 M lead(II) nitrate.The tubes on the second row contain 0.1 M copper(II) nitrate. The reactivity series of metals is a list of metals arranged in their order of reactivity from highest to lowest. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
